Ph3 molecular geometry4/15/2023 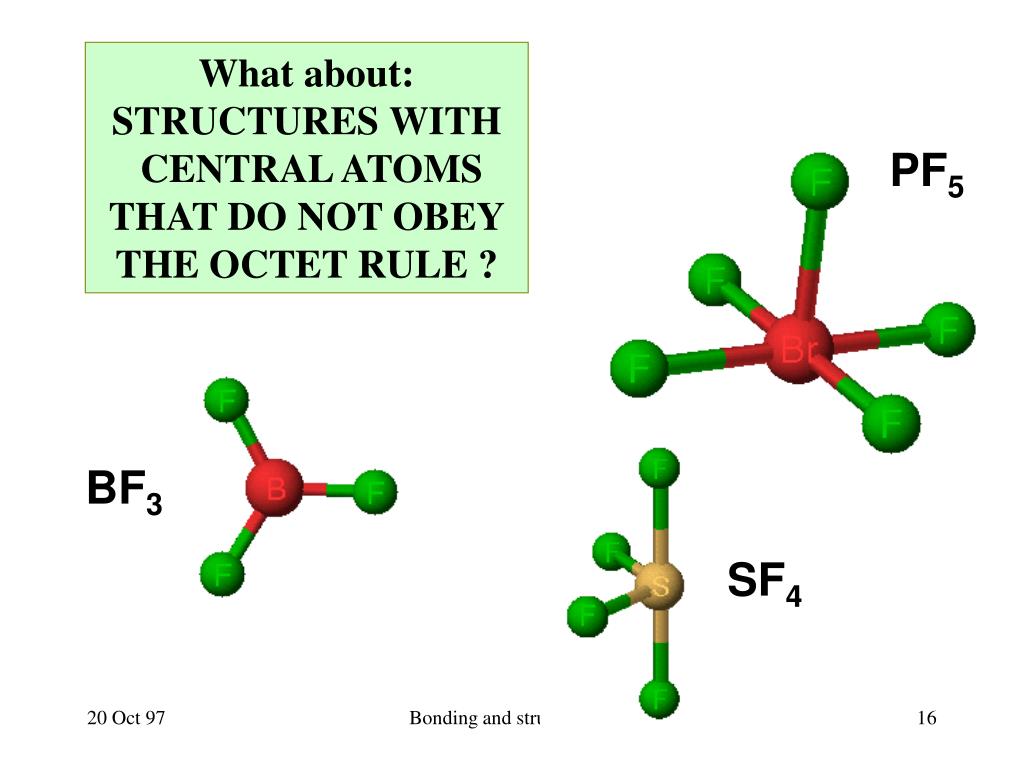
What intermolecular forces are present in PH3? If these are all bond pairs the molecular geometry is tetrahedral (e.g. See also How can I measure 3 ounces? What is the molecular geometry of CH4?įor example four electron pairs are distributed in a tetrahedral shape. So essentially it is only applicable for PH3, AsH3,SbH3 and H2S, H2Se, H2Te. What is a Drago molecule?ĭrago’s rule is an empirical rule and is used to explain the bond angles of hydrides of group 15 and 16 of or below 3rd period. There for more bond angle also in PH3 no backbonding can take place as hydrogen has no pair ofelectron(e) for backbonding to take place. Drago molecules do not have any hybridisation and have the least bond angles. But in reality, PH3 molecule has no hybridisation. 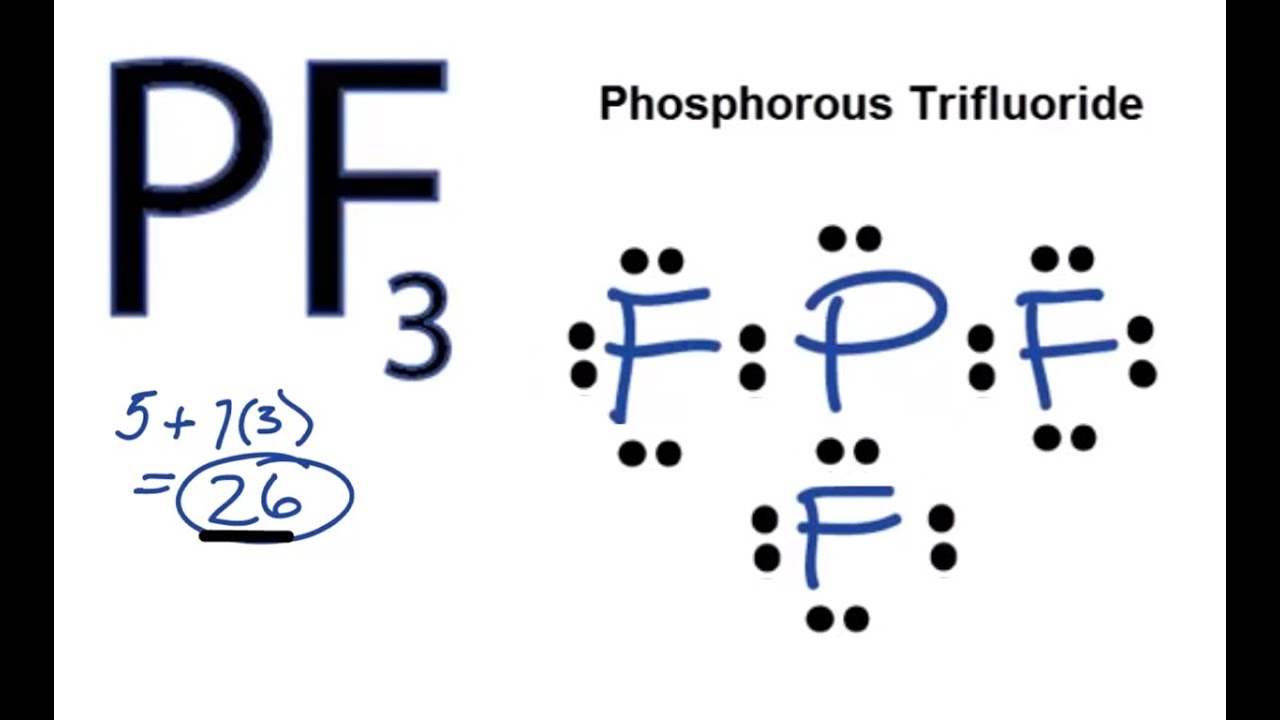
The hybridisation in PH3 molecule seems like sp3 according to its Lewis structure and Steric number rule. Phosphorus’s electronegativity is a nonpolar molecule because it is the same, but since Phosphorus has a lone pair, PH3 is a polar molecule. PH3 is a polar molecule because it has a bent structure due to lone pairs of electrons and electron-electron repulsion. This results in large repulsion between P-F bonds and hence bond angle is large. But PF3 has partial double bond character due to back π-donation of electrons from p orbitals of F atom to empty d orbitals of P. Why bond angle of PH3 is less than PF3?īoth PH3 and PF3 are pyramidal in shape. Hence the bond angle PH+4 in is higher than the bond angle in PH3. Due to the absence of lone pair in PH+4 there is no lone pair-bond pair repulsion. PH3 combines with a proton to form PH+4 in which the lone pair is absent. See also Does Neji marry Tenten? Why bond angle of PH3 is less than PH4? There are two S-O double bonds at the SO2 molecular geometry. The SO2 molecule has a V-shaped or bent geometry because it contains two oxygen atoms in the V-shaped or bent form and two corners with one lone pairs of electrons on central sulfur atom. What is the molecular geometry of PH3? When you have 2 electron groups and none are lone pairs, the molecular geometry is linear. What is the molecular geometry of PH3 quizlet? Which of the following has a tetrahedral electron geometry and a trigonal pyramidal molecular geometry?.What are the angles in a trigonal planar geometry?.What electron geometry is associated with sp2 hybridization?.What intermolecular forces are present in PH3?.Why bond angle of PH3 is less than PF3?.Why bond angle of PH3 is less than PH4?.What is the molecular geometry of a CO 2 molecule quizlet?.What is the molecular geometry of PH3 quizlet?.Both electron domains are bonding pairs, so CO2 has a linear molecular geometry with a bond angle of 180°. What is geometry of CO2?ĬO2 has 2 electron domains, resulting in a linear electron domain geometry. The central atom oxygen creates a single bond with the two hydrogen atoms. What is the molecular geometry of h2o enter the molecular geometry of the molecule?Īnswer and Explanation: The molecular geometry of water is bent. With this, the PH3 is a polar molecule with nonpolar covalent bonds, not polar bonds. 
However, due to the unbonded electron, there will be asymmetrical charge distribution. The electronegative property of PH3 found in the periodic table attracts shared pairs of valence electrons, creating covalent bonds. Answer: The PH3 P H 3 molecule is (d) Trigonal bipyramidal. To find the structure of PH3 P H 3 molecule. Molecular geometry around phosphorous atom is tetrahedral. No charges on phosphorous atom and hydrogen atoms. In phosphine (PH 3) lewis structure, there are three sigma bonds and one lone-pair around phosphorous atom. Are you looking for the answer to the question: What Is the Molecular Geometry of Ph3 Enter the Molecular Geometry of the Molecule? We have gathered for you the most accurate and comprehensive information that will fully answer the question: What Is the Molecular Geometry of Ph3 Enter the Molecular Geometry of the Molecule? Is PH3 trigonal pyramidal?
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
